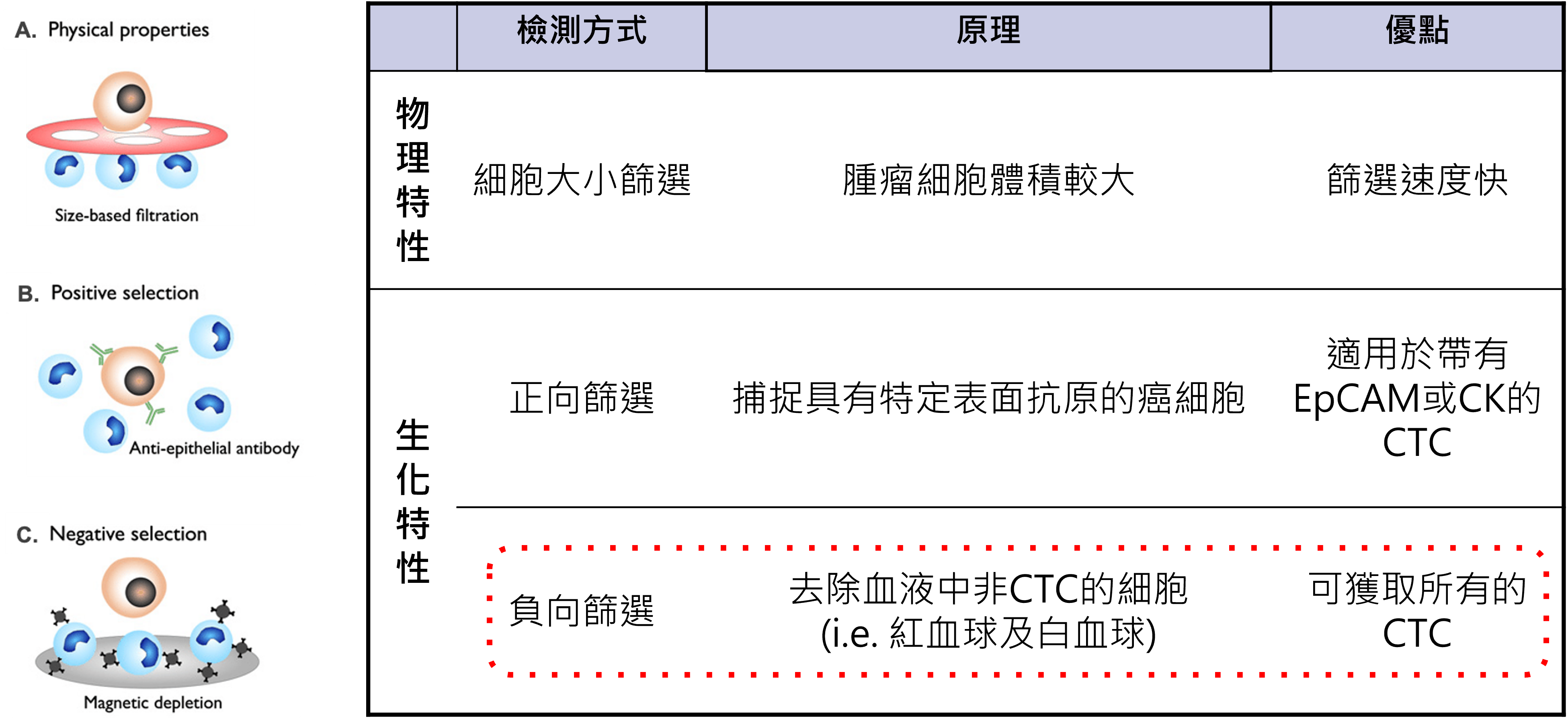
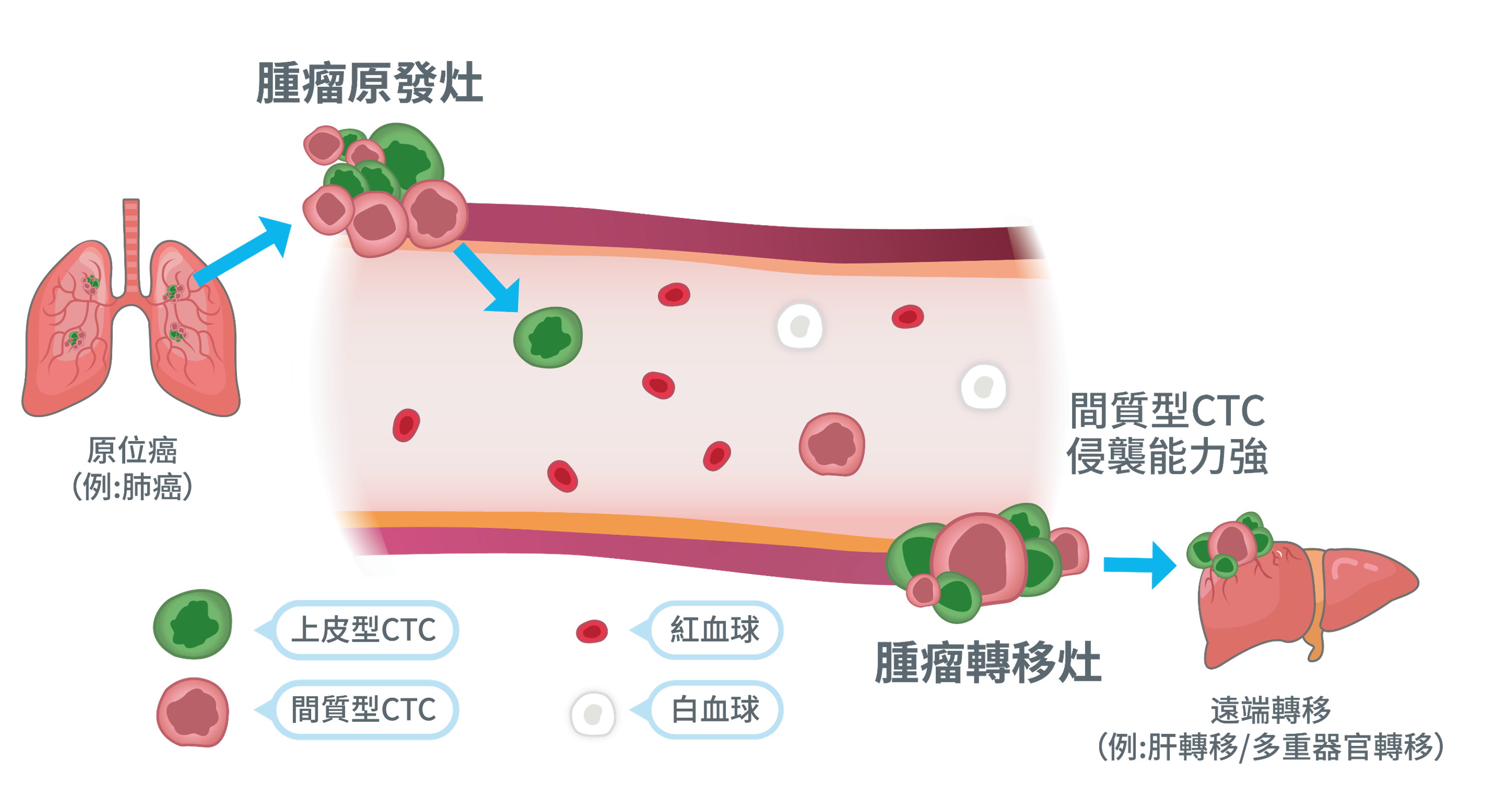
circulating tumor cells can be roughly divided into two paths that rely on physical properties and those that rely on biological properties. The former uses differences in cell size and chargeability for separation, while the latter identifies based on surface protein expression. The problem is that tumor cells themselves are highly heterogeneous, and if you only rely on a single condition for capture, there is often a risk of missing out.
The so-called forward screening is to first find cells that match specific tumor markers and capture them. This method is relatively straightforward when the marker expression is stable, but once the tumor cells undergo phenotypic changes during the metastasis process, or are not originally a population that expresses the marker highly, it may not be fully retained. The enrichment methods of
The core idea adopted by Good Future is negative screening. The actual process will first use the difference in osmotic pressure to remove red blood cells, then use specific antibodies to identify white blood cells, and use the PowerMag magnetic column to remove interfering blood cells from the blood, retaining non-blood cells as much as possible. This kind of design does not first assume that tumor cells must grow in a certain way, but first eliminates unnecessary cells and then retains more targets that may be meaningful.
When immunofluorescence staining, automated microscopic photography, and cell image interpretation are subsequently added, the advantages of negative screening are truly amplified. It allows the back-end report to not only be determined by a single signal, but to retain more complete cell images, staining combinations and interpretation basis, reducing leakage and misjudgment caused by only looking at a single marker.